Classification and staging of Mycosis Fungoides and Sézary Syndrome involves evaluation of 4 disease compartments (TNMB)1
Using TNMB criteria, each compartment is classified according to the extent and type of disease involvement in skin, lymph nodes, viscera, and blood.
| CLINICAL STAGE | CLASSIFICATIONa | |||||
|---|---|---|---|---|---|---|
| T Skin |
N Lymph Nodes |
M Metastasis |
B Blood |
|||
| Limited skin involvement | IA | T1 | N0 | M0 | B0-1 | |
| Skin-only disease | IB | T2 | N0 | M0 | B0-1 | |
| IIA | T1-2 | N1-2 | M0 | B0-1 | ||
| Tumor stage | IIB | T3 | N0-2 | M0 | B0-1 | |
| Erythrodermic stage | IIIA | T4 | N0-2 | M0 | B0 | |
| IIIB | T4 | N0-2 | M0 | B1 | ||
| Sézary Syndrome (SS) | IVA1 | T1-4 | N0-2 | M0 | B2 | |
| SS or Mycosis Fungoides (MF) |
IVA2 | T1-4 | N3 | M0 | B0-2 | |
| Visceral disease | IVB | T1-4 | N0-3 | M1 | B0-2 | |
T1=Limited patches/plaques, <10% of skin surface
T2=Generalized patches/plaques, ≥10% of skin surface
T3=≥1 tumor(s) (≥1 cm in diameter)
T4=Erythroderma, ≥80% of skin surface
N0=No abnormal nodes
N1=Clinically abnormal histopathology, Dutch Grade 1 or NCI-LN0-2
N2=Clinically abnormal histopathology, Dutch Grade 2 or NCI-LN3
N3=Clinically abnormal histopathology, Dutch Grade 3-4 or NCI-LN4
M0=No visceral involvement
M1=Visceral involvement
B0=No blood involvement (<250 atypical cells/μL of lymphocytes)
B1=Low-level involvement (250-1000 atypical cells/μL of lymphocytes)
B2=High-level involvement (>1000 atypical cells/μL of lymphocytes)
Be vigilant for signs of progression in your patients with Mycosis Fungoides and Sézary Syndrome
About 1 in 3 patients with Mycosis Fungoides may experience progression within skin or to other compartments2,b
Up to 20% of patients with early-stage Mycosis Fungoides may have blood involvement3,c

More extensive skin involvement is associated with poor outcomes2,b
Overall survival, by T classification2
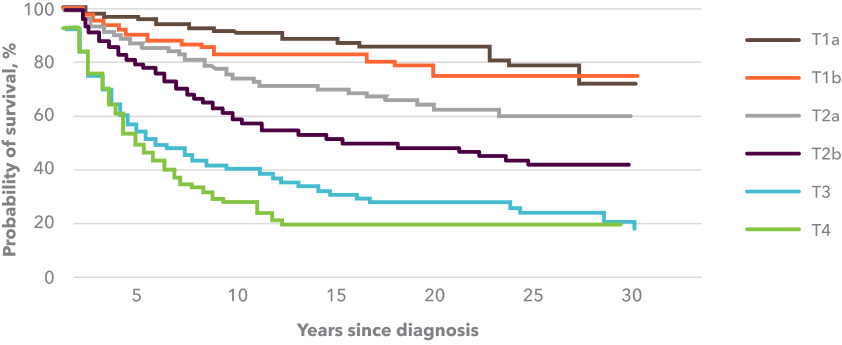
Adapted from Agar N, Wedgeworth E, Crichton S, et al. Journal of Clinical Oncology, Volume 28, Issue 31, p4730-4739. American Society of Clinical Oncology.
- The considerable pruritus and pain caused by skin lesions, patches, and plaques of Mycosis Fungoides are associated with increased fatigue, anxiety, and depression3,4
- These symptoms can have a substantial negative impact on daily function and emotional/social well-being3,4
Blood testing via flow cytometry for suspected MF or SS is important at diagnosis and throughout treatment to monitor disease burden and response to treatment1

- aBlood, viscera, and lymph node classification for each level can further be characterized by T-cell receptor clone status, with A=clone negative or equivocal and B=clone positive with identical clone in the skin.1,5
- bBased on a survival outcomes and prognostic factors study that included 1502 patients with Mycosis Fungoides/Sézary Syndrome.2
- cBased on a PROCLIPI database analysis that included 348 patients with early-stage Mycosis Fungoides.3
- CCR4=C-C chemokine receptor type 4; NCI-LN=National Cancer Institute - Lymph Nodes grade system; TNMB=tumor-node-metastasis-blood
See how POTELIGEO targets CCR4+
T cells for destruction
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Primary Cutaneous Lymphomas V.1.2025. © National Comprehensive Cancer Network, Inc. 2024. All rights reserved. Accessed February 11, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org.
- Agar NS, Wedgeworth E, Crichton S, et al. Survival outcomes and prognostic factors in mycosis fungoides/Sézary syndrome: validation of the revised International Society for Cutaneous Lymphomas/European Organisation for Research and Treatment of Cancer staging proposal. J Clin Oncol. 2010;28(31):4730-4739.
- Scarisbrick JJ, Quaglino P, Prince HM, et al. The PROCLIPI international registry of early-stage mycosis fungoides identifies substantial diagnostic delay in most patients. Br J Dermatol. 2019;181(2):350-357.
- Ottevanger R, van Beugen S, Evers AWM, Willemze R, Vermeer MH, Quint KD. Quality of life in patients with mycosis fungoides and Sézary syndrome: a systematic review of the literature. J Eur Acad Dermatol Venereol. 2021;35(12):2377-2387.
- Olsen EA, Whittaker S, Willemze R, et al. Primary cutaneous lymphoma: recommendations for clinical trial design and staging update from the ISCL, USCLC, and EORTC. Blood. 2022;140(5):419-437.
